▼This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Please report any adverse events via your national reporting system. Adverse events can also be reported to Alexion Pharmaceuticals by contacting: https://contactazmedical.astrazeneca.com.
Please refer to the Summary of Product Characteristics for further information.

healthcare professional
Image created for Alexion Pharmaceuticals, Inc. for illustrative purposes.
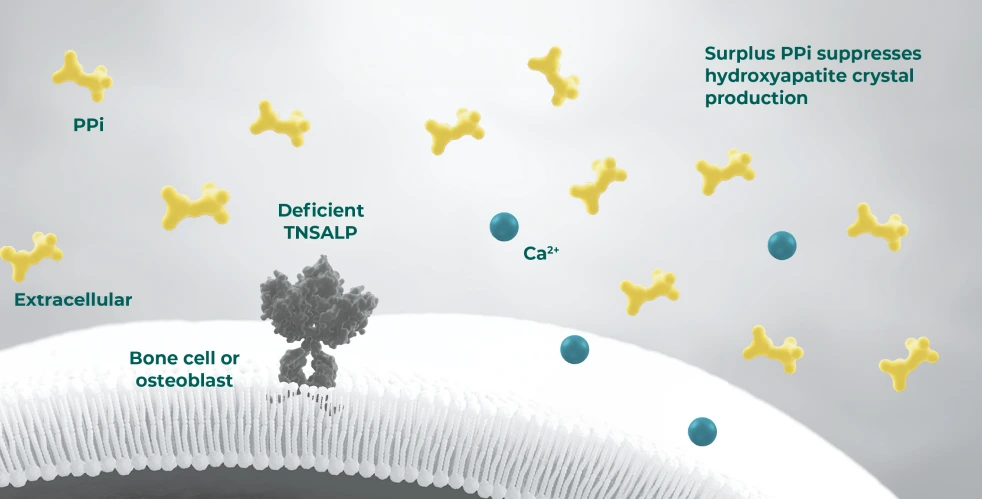
- In HPP, deficiency in the activity of the enzyme TNSALP leads to harmful accumulation of substrates PPi and PLP, impairing bone mineralisation and vitamin B6 metabolism, respectively8–11
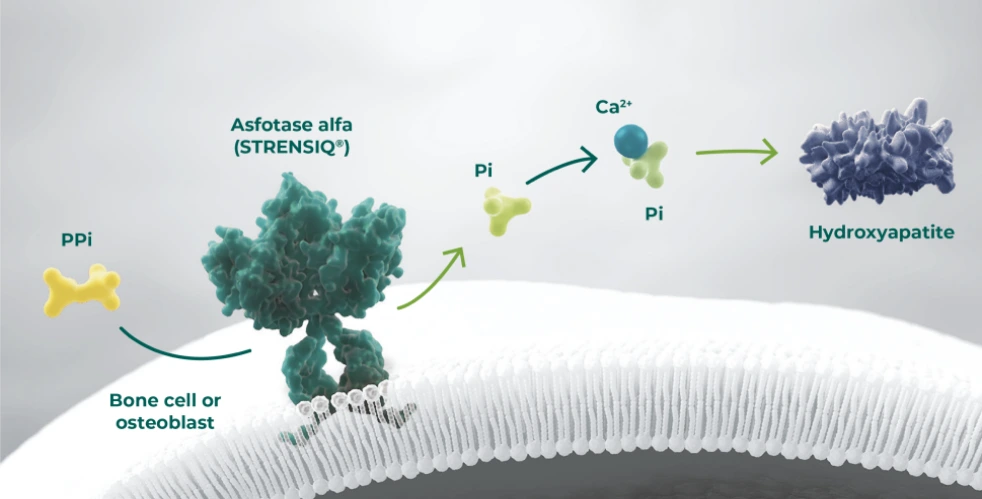
- By replacing TNSALP, STRENSIQ® may promote bone mineralisation and a decrease in elevated TNSALP substrate levels, namely PPi and PLP1,6,7,11
- With STRENSIQ®, the median serum ALP activity can rise well above the upper limit of normal (age and sex dependent normal range) and may be sustained over time with subcutaneous dosing2,6

ALP, alkaline phosphatase; PLP, plasma/serum pyridoxal 5′-phosphate; Pi, inorganic phosphate; PPi, inorganic pyrophosphate; TNSALP, tissue non-specific alkaline phosphatase.
This medicinal product has been authorised under ‘exceptional circumstances’. This means that due to the rarity of the disease it has not been possible to obtain complete information on this medicinal product.
▼This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Please report any adverse events via your national reporting system. Adverse events can also be reported to Alexion Pharmaceuticals by the following link: https://contactazmedical.astrazeneca.com/
▼This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Please report any adverse events via your national reporting system. Adverse events can also be reported to Alexion Pharmaceuticals by the following link: https://contactazmedical.astrazeneca.com/